The Science Behind Our Topical Muscle Relaxant with Antispasmodic Action
Introduction to Our Scientific Approach
Prima Innovations LLC is advancing a first-in-class topical muscle relaxant with antispasmodic action designed to act directly at the site of muscle spasm. Our scientific platform is built on the principle that effective muscle spasm treatment should target the affected skeletal muscle tissue itself, rather than relying primarily on systemic central nervous system (CNS) mechanisms.
Prima Innovations is developing a differentiated pharmaceutical solution — one that focuses on localized pharmacologic action within muscle tissue. Our goal is to create a muscle relaxant drug that delivers a measurable antispasmodic in 10 minutes or less directly where abnormal muscle contraction is present without any unwanted side effects.
Targeting Muscle Spasms at the Source
Muscle spasms occur when skeletal muscle fibers contract involuntarily and fail to relax properly. These spasms may result from acute injury, chronic strain, neurological signaling imbalance, or inflammatory processes. In many cases, muscle hyperactivity contributes significantly to pain and functional limitation.
Prima Innovations’ investigational therapy is formulated for topical administration and designed to penetrate dermal layers to reach underlying skeletal muscle tissue. By targeting muscle pathways locally, our therapy is intended to:
- Support antispasmodic muscle pain relief
- Interrupt abnormal muscle contraction cycles
- Provide localized muscle spasm treatment
- Offer a differentiated mechanism compared to centrally acting drugs
This localized approach represents a meaningful scientific shift in the development of medication for painful muscle spasms.
Pharmaceutical-Grade Topical Innovation
Unlike over-the-counter creams or counterirritant products that primarily mask discomfort, Prima Innovations’ therapy is being developed as a pharmaceutical-grade skeletal muscle relaxant. The formulation strategy prioritizes:
- Controlled dermal penetration
- Targeted delivery to muscle tissue
- Stability and reproducibility under pharmaceutical manufacturing standards
- Regulatory-aligned development practices
Our drug development program integrates pharmacology, formulation chemistry, and translational research to support a robust clinical pathway.
The investigational compound is engineered to exhibit antispasmodic activity within muscle tissue without relying primarily on central nervous system sedation mechanisms. By limiting systemic exposure, our approach may offer a differentiated profile compared to traditional oral muscle relaxant drugs.
Preclinical Foundation and Translational Research
Prima Innovations has completed preclinical evaluations designed to establish proof-of-concept for its topical muscle relaxant platform. In relevant experimental models of muscle spasm and contraction, the investigational therapy demonstrated promising pharmacodynamic activity.
Preclinical development has focused on:
- Mechanistic validation of localized muscle relaxation
- Evaluation of dermal absorption and tissue distribution
- Assessment of pharmacologic response in muscle tissue
- Safety and tolerability profiling in early studies
These data support continued advancement toward formal clinical development.
As with all pre-clinical stage pharmaceutical programs, safety and efficacy must be established through carefully designed human trials and regulatory review. Prima Innovations remains committed to scientific rigor as it progresses through development milestones.
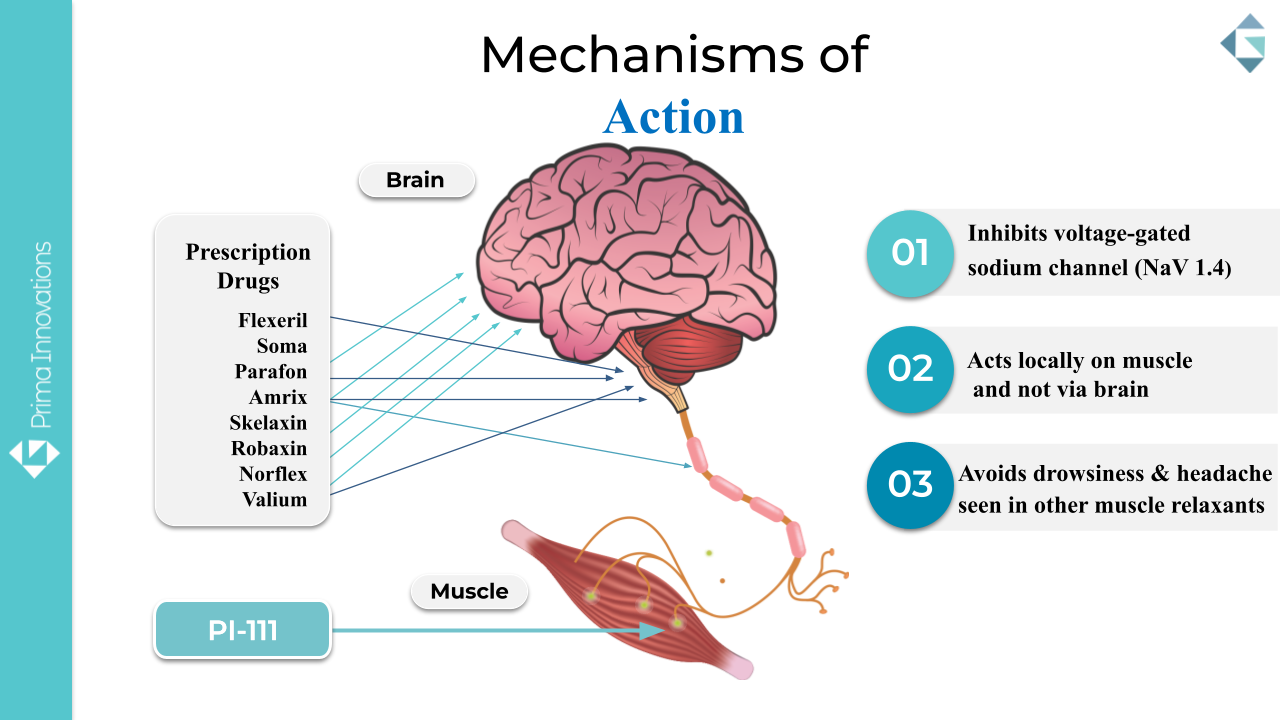
Differentiation from Traditional Muscle Relaxant Medications
Most currently prescribed muscle relaxant medications act through central neurological pathways. While these therapies may reduce muscle tone, they may also be associated with systemic side effects due to widespread CNS activity.
Prima Innovations is developing a fast-acting muscle relaxant formulated for localized action. Key areas of differentiation include:
- Topical administration rather than oral systemic dosing
- Muscle relaxation in 10 minutes or less without systemic side effects
- Multi-layered IP strategy
- Efficient and cost effective approval pathway
By addressing muscle contraction at the tissue level, our approach may represent an evolution in how back pain muscle relaxant therapies and neck pain muscle spasm relief options are developed.
Intellectual Property and Platform Potential
Prima Innovations has secured patent protection covering aspects of its topical antispasmodic medication platform. Our intellectual property strategy is designed to protect:
- Method of use
- Combination products
- Future pipeline extensions
This patent-protected foundation provides strategic value for investors and development partners seeking differentiated assets within the musculoskeletal and pain management markets.
Beyond the lead indication, our localized muscle-targeting technology may support expansion into additional therapeutic areas involving muscle hyperactivity or spasticity treatment medication needs.

Commitment to Scientific Integrity
At Prima Innovations, scientific credibility and regulatory alignment are central to our mission. As a pre-clinical stage pharmaceutical company, we are committed to:
- Transparent communication of development status
- Data-driven advancement through clinical milestones
- Compliance with FDA and global regulatory frameworks
- Responsible representation of investigational outcomes
Our development program is structured to meet the expectations of regulatory authorities, institutional investors, and strategic pharmaceutical partners.
Frequently Asked Questions
Q:
How does this therapy differ from common topical pain creams?
Most topical creams provide temporary sensation-based relief. Prima Innovations’ therapy is being developed as a true antispasmodic medication designed to act pharmacologically within muscle tissue.
Q:
Is the product currently approved?
No. Our topical muscle relaxant drug is in clinical-stage development and has not yet received regulatory approval.
Q:
What types of muscle conditions could this address?
The investigational program is focused on muscle spasm treatment, acute musculoskeletal pain relief, back pain muscle relaxant needs, neck pain muscle spasm relief, and potential spasticity-related applications.
Q:
What is the next milestone?
Prima Innovations is advancing regulatory and clinical planning activities to support progression into formal human studies.
Partner with Prima Innovations
Prima Innovations LLC is building a next-generation approach to antispasmodic muscle pain relief through localized pharmaceutical innovation. We welcome conversations with investors, research collaborators, and strategic partners who share our vision of redefining muscle spasm treatment.
Thank you for contacting us.We’ll get back to you as soon as possible.
We got it.
Ready to begin?
You don’t have to take the next step alone.
Reaching out is the first step.


